|
9/2/2023 0 Comments Properties of physical change 
In other words, the extensive property of the total system is the sum of extensive properties of every subsystem. An extensive property is the additive of subsystems. Thus, the volume is an extensive property. One litre of a bottle holds one-litre water and two litres of a bottle holds two-litre water. An extensive property is directly proportional to the amount of a substance. Extensive PropertiesĮxtensive properties are which depend on the amount of a substance or the size of a system. Some of the examples of intensive properties are density, colour, temperature, conductivity, refractive index, malleability, lustre, hardness etc. So, the density of water always will be 1000 kg m −3 irrespective of the mass and/or volume of water. The density of water is 1000 kg m −3, and it is an intensive property. Intensive properties are the same for 1 gram of a substance or 1 kilogram, 1 cm 3 or 1 m 3. The property does not change with the size of a system. Intensive PropertiesĪ property of a substance or system is called intensive when it is independent of the amount of the substance. Other examples of chemical change are milk turning sour, burning of paper, combustion of petrol or any other fuel, fermentation.Įxample of chemical change (clockwise from top left: digestion, photosynthesis, firefox, nuclear bombing, combustion, and rusting) 
The chemical composition of the original material is completely altered. The original iron is permanently destroyed to produce iron oxide. In corrosion, iron reacts with oxygen to form iron oxide. Chemical properties of a substance are indications of the chemical nature of the substance like reactivity towards acids and bases, combustibility, flammability, acidity, basicity.Ĭhemical change alters the chemical composition of a substance. Substances whose properties are to be estimated are lost during the measurement. The chemical change is nothing but a chemical reaction. It means a substance changes its original identity during the measurement of its chemical properties. Chemical PropertiesĬhemical properties can only be estimated through a chemical change. 
The chemical composition of a substance remains fixed during the measurement of physical properties in other words, there is no chemical change.Įxamples of physical change (clockwise from top left: drying, ripped paper, melting, chopped wood, and distillation) Physical properties can be measured or determined without altering the chemical identity of a substance. 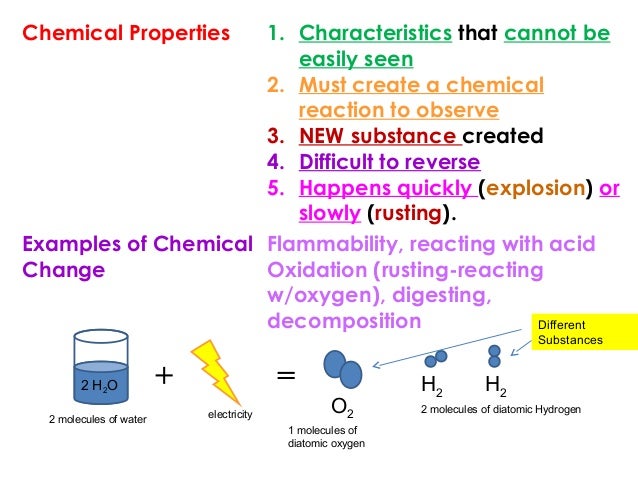
Properties of Matter (Chemical and Physical, Intensive and Extensive) Physical Properties Based on the dependence of properties on the amount of a substance, we can classify them into intensive properties and extensive properties. Based on how properties are determined, they can be classified into two categories: physical properties and chemical properties. There are two ways in which we can classify properties of matter. Some of these properties can merely be identified by simple observation while some require experimentation. With the knowledge of properties of substances, chemists can predict their behaviour, nature, and their interactions with other substances. These characteristics and attributes are the properties of matter. All Substances behave differently based on their characteristics and attributes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
